Turning clinical insight into manufacturable science.
Akrivis Health Care builds the materials, devices, and biological systems that the next decade of medicine will need. Our platforms span synthetic exosome-mimetic therapeutics, catechol-melanin tissue adhesives, and marine haemoglobin oxygenation systems — engineered in India, designed for the world.
We build platforms, not products. Each product is the expression of a platform; each platform is a body of knowledge that compounds.
Dr. Sarat Babu Janipalli
·Founder & Managing Director
Five platforms. Each a body of knowledge that compounds.
Akrivis organises its work into platforms — not products. A platform is a body of knowledge, intellectual property, and manufacturing capability that can yield several products. The platform story is what makes Akrivis a deep tech company rather than a contract manufacturer.
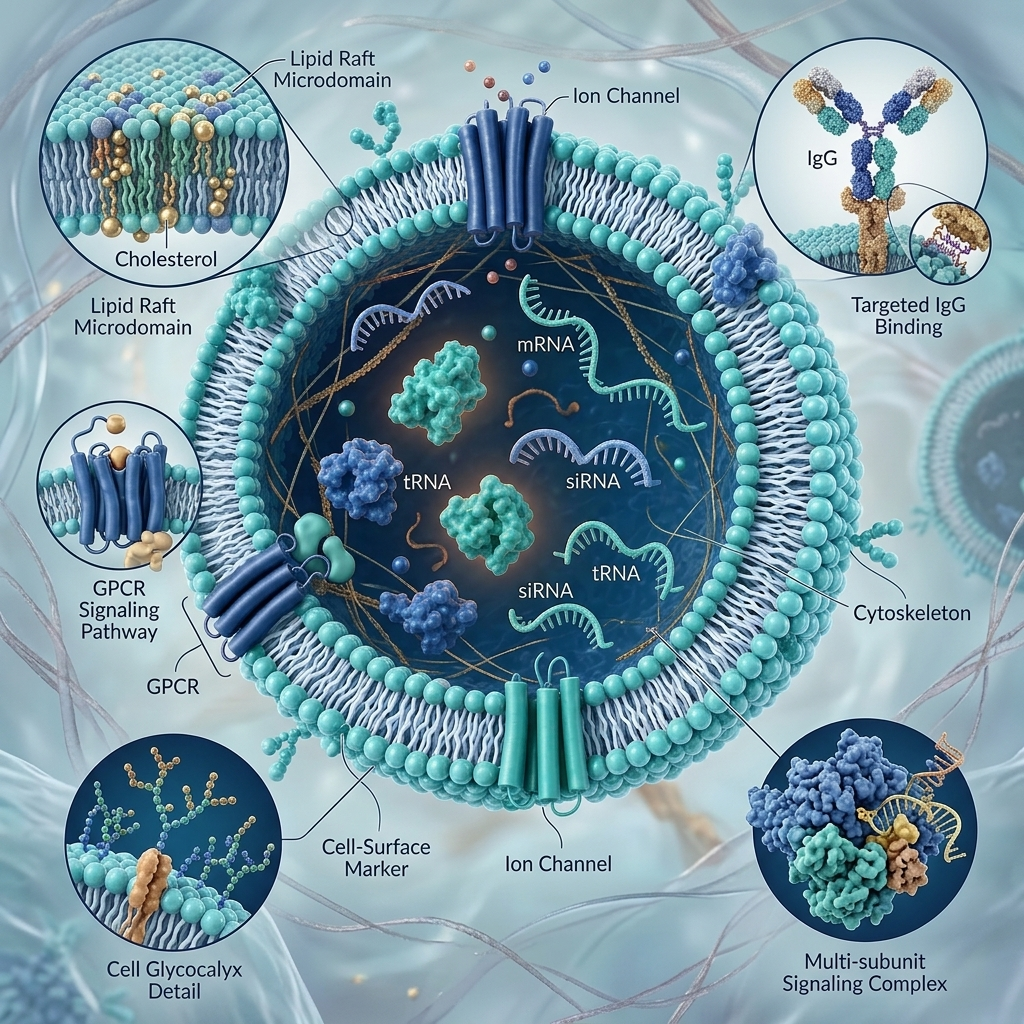
SynExo™ Synthetic Exosome-Mimetic Nano-Vesicle — Structural ArchitectureHover or tap the markers to explore each component
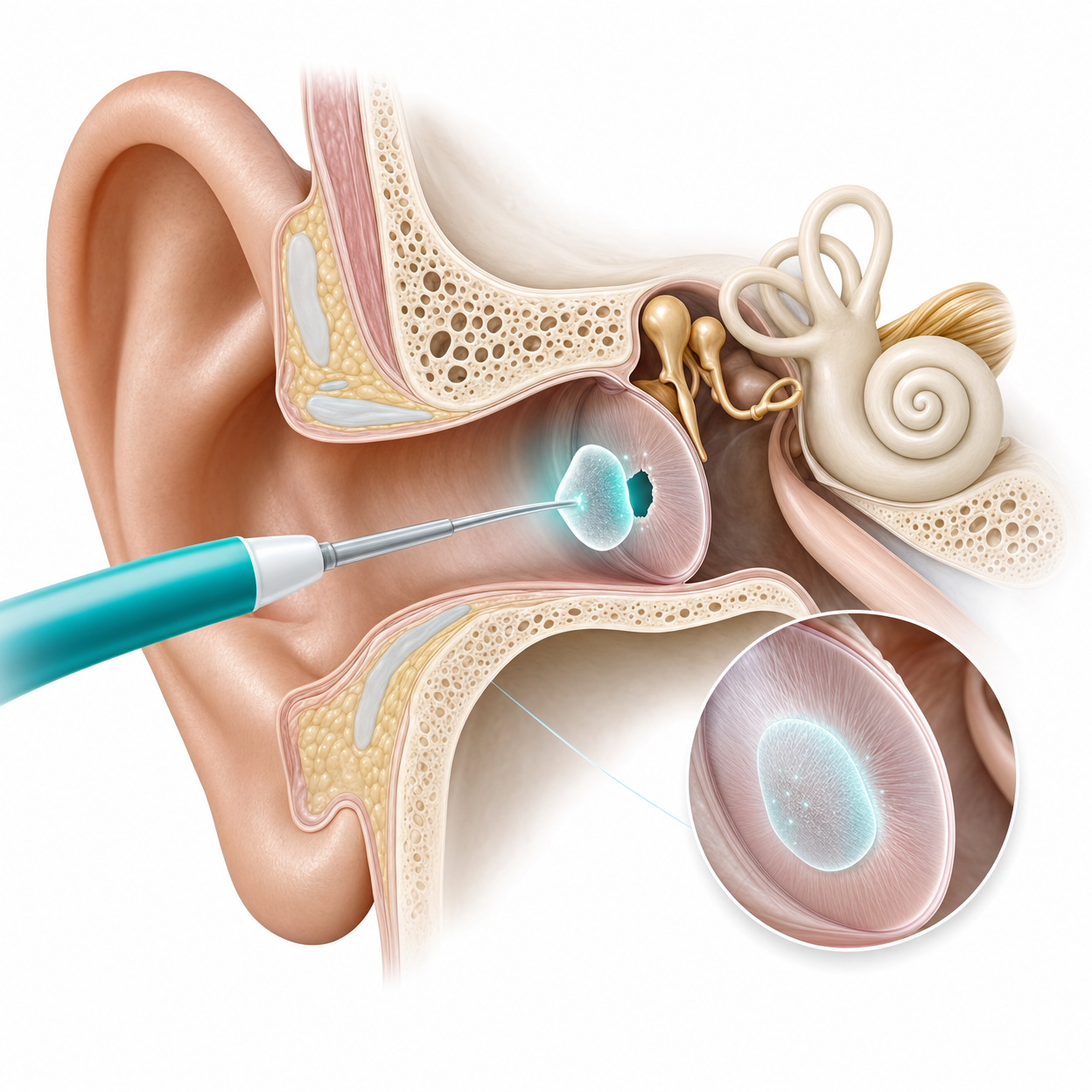
TympanoSeal™ NeoTympanum — Non-Invasive Tympanic Membrane RepairCatechol-melanin bioadhesive on PLA scaffold, activated by pulsed 9.0 J/cm² light
Catechol-Melanin Adhesion
TympanoSeal™ NeoTympanum
Dual-mechanism tissue adhesive exploiting mussel-inspired catechol bioadhesion paired with the Raper–Mason melanin pathway. Lyophilised seven-component formulation on a PLA scaffold, activated by pulsed 9.0 J/cm² light. The world’s first non-invasive alternative to tympanoplasty surgery — a fifteen-minute in-clinic procedure targeting the 10M+ Indians affected annually.
SynExo™ Therapeutics
Synthetic Exosome-Mimetic Nano-Vesicles
Synthetic bioactive peptides (copper tripeptide, palmitoyl pentapeptide) encapsulated in exosome-mimetic nano-vesicles (30–150 nm). Mimics natural exosome regenerative signalling without live-cell complexity. The synthetic peptide backbone carries an established CDSCO safety profile, enabling an accelerated regulatory pathway.
EXOGLAM
Exosomes & Plant-Derived Exosome-Like Nanoparticles
Therapeutic and cosmetic formulations built on exosomes and plant-derived exosome-like nanoparticles (PELNs). Applications span aesthetic medicine, wound repair, and adjacent regenerative indications. European market access strategy supports international expansion.
HEMOSOME OxyRescue Serum
Marine Haemoglobin — Hemarina SA, France
Commercialisation of the OXYMARIS / Hemosome technology developed by Hemarina SA of France. Marine haemoglobin extracted from Arenicola marina carries oxygen across mammalian biological systems. Topical oxygenation applications in wound healing and aesthetic medicine.
ClearSight Nano · Nano Collagen · VOC · MPTR
Nanomaterials, Diagnostics & Epigenetic Reprogramming
ClearSight Nano: nanomaterials platform with optical applications. Nano Collagen: exclusive supply with Holista Colltech (ASX: HCT), signed December 2025. VOC breathalyzer: diagnostic device exploiting volatile organic compound signatures. MPTR: autologous epigenetic reprogramming delivering Yamanaka factors via targeted nanoparticle systems.
From validated cosmeceuticals to therapeutic pipeline.
A validated cosmeceutical portfolio supports the development of regenerative therapeutics and first-in-class medical devices. Each product leads with the clinical problem it solves, followed by the mechanism, then the evidence, then the development stage.
Therapeutic Pipeline

SynExo™ Wound Gel
Chronic & acute wound healing
Exosome-mimetic nano-vesicles delivering copper tripeptide + palmitoyl pentapeptide

SynExo™ Burn Gel
Partial-thickness burn management
Nano-vesicle encapsulated regenerative peptides for dermal remodelling
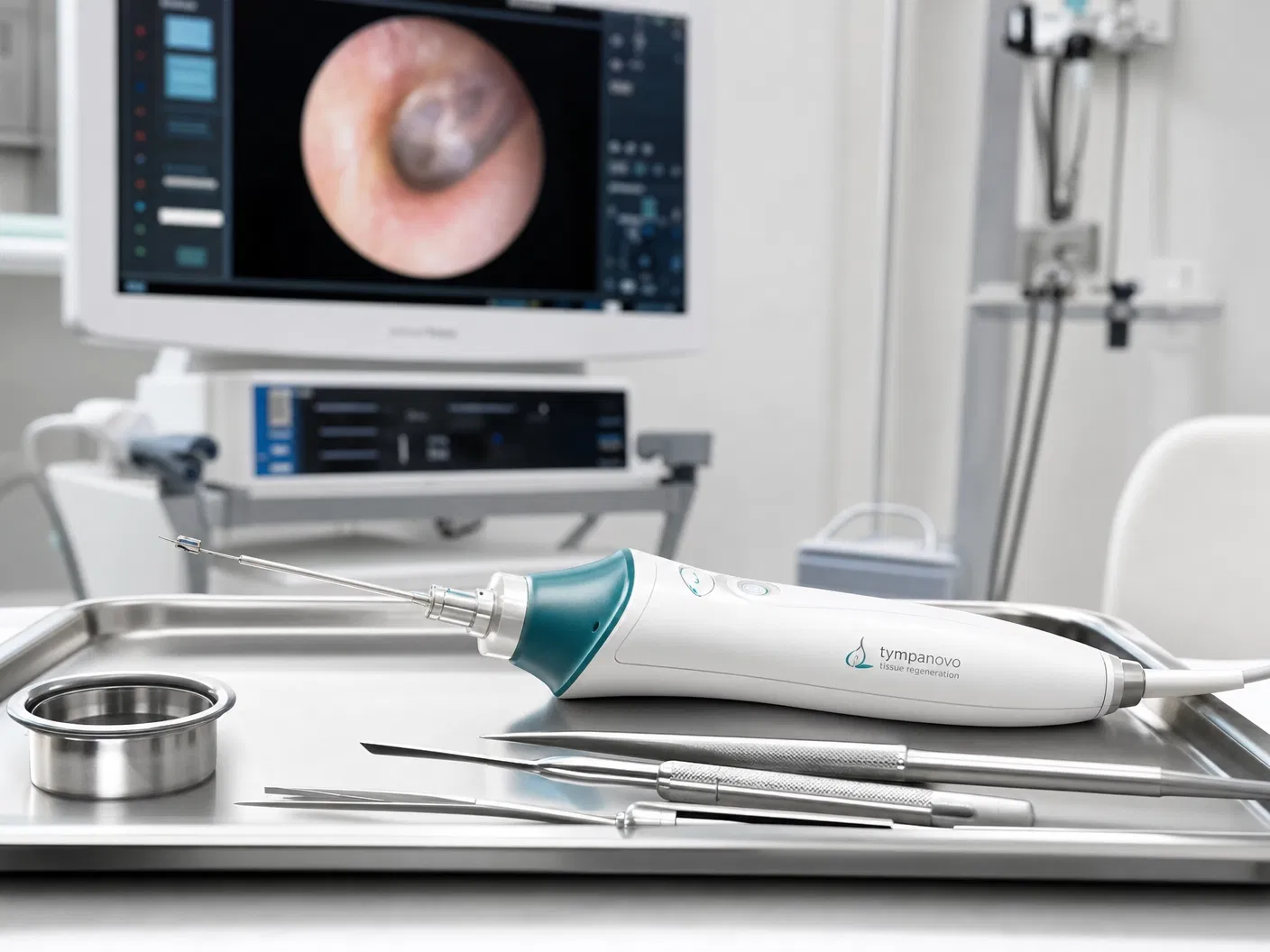
TympanoSeal™ NeoTympanum
Tympanic membrane perforation repair
FMN–catechol cross-linking + Raper–Mason melanogenesis on PLA scaffold


Commercial Portfolio
EXOGLAM Serums
Aesthetic Medicine
CDSCO-Approved
Nano Collagen Range
Dermal Remodelling
Holista Colltech (ASX: HCT) Exclusive Supply
HEMOSOME OxyRescue
Topical Oxygenation
Hemarina SA Partnership
SynExo™ Hair Growth
Trichology
CDSCO-Approved
SynExo™ Anti-Ageing
Cosmeceutical
CDSCO-Approved
Built by scientists, scaled by operators.
Seven founders and directors with 85+ combined years across nanomaterials, clinical research, regulatory affairs, international business development, and strategic finance.
Dual-Entity Structure
Akrivis Health Care Private Limited is the commercial operating entity — manufacturing, sales, and distribution. HCRB (Human Cell Research Bharat) is the IP vault and R&D entity that owns the patent portfolio and licenses technology to Akrivis. This structure separates commercial risk from intellectual property.
Dr. Sarat Babu Janipalli
Founder & Managing Director
Clinical to formulation to systems — scientist with two decades of experience in surgery with interests in nanomaterials and regenerative medicine. Leads platform architecture, IP strategy, and regulatory navigation. Architect of the SynExo™ manufacturing process.
Dr. Vamshi Krishna Irlapati
Co-Founder & Chief Scientific Officer
Leads pre-clinical research, assay development, and scientific validation across all six platforms. Translates platform science into reproducible manufacturing protocols and regulatory-grade documentation.
Mr. Ravi Eswarapu
Co-Founder & Chief Business Officer
Drives commercial strategy, international partnerships, and investor relations. Architect of the B2B distribution network spanning India, Europe, and the Middle East.
Mr. Sampath Kumar Janipalli
Founder — International Operations
Manages international operations, entity coordination, and cross-border supply chain logistics. Anchors European distribution and partnership infrastructure.
Mr. Abhijit Samant
Director — Regulatory Affairs & Quality Systems
Oversees CDSCO submissions, ISO compliance, and quality management systems across the AMTZ manufacturing facility. Navigates accelerated regulatory pathways for therapeutic approvals.
Mr. Kuldeep Singh
Director — Manufacturing & Supply Chain
Leads manufacturing operations at the AMTZ facility, supply chain optimisation, and production scaling. Maintains cGMP compliance and quality standards across the commercial portfolio.
CA (Dr.) Mahaveer Chaplot
Founder — Private Equity & Strategic Finance
Prominent Chartered Accountant, educator, and community leader with over two decades of expertise in private equity, banking, and agripreneurship. Director of Agribid, an AI-driven platform empowering farmers. General Secretary of the Certified Accountants Division, International Council of Jurists. Currently serving as Sachiv (Secretary) for JITO Apex (2024–26).
Compliance is infrastructure, not a checkbox.
Akrivis maintains active certifications across Indian and international regulatory frameworks. The AMTZ facility in Visakhapatnam serves as the manufacturing anchor, operating under cGMP protocols with continuous quality monitoring.
CDSCO
Central Drugs Standard Control Organisation
Manufacturing licence for cosmeceutical and therapeutic formulations. Accelerated regulatory pathway for synthetic peptide-based therapeutics. Full commercial portfolio approved.
ISO 22716:2007
Cosmetics GMP
Good Manufacturing Practice for cosmetic products. Covers the AMTZ facility production lines for the entire EXOGLAM and SynExo™ cosmeceutical portfolio.
ISO 9001:2015
Quality Management Systems
Organisation-wide quality management system certification covering design, development, manufacturing, and distribution of cosmeceutical and therapeutic products.
US FDA
United States Food & Drug Administration
Facility registration and product listing for export-grade cosmeceutical formulations. Enables direct export through registered distribution channels.
BIS MHD-20
Bureau of Indian Standards — Medical & Healthcare Devices
Institutional membership on the MHD-20 committee. Akrivis participates in drafting Indian standards for exosome-based products and medical devices. This is an institutional credential.
TÜV Rheinland
Independent Elemental Validation
Independent third-party validation of platform materials via Inductively Coupled Plasma Mass Spectrometry. Confirms elemental composition and purity across the SynExo and EXOGLAM portfolios.
Manufacturing Anchor: AMTZ, Visakhapatnam
The Andhra Pradesh MedTech Zone (AMTZ) is Asia's first integrated medical device manufacturing ecosystem. Akrivis operates its cGMP facility within AMTZ, providing access to shared analytical infrastructure, regulatory support, and a cluster of complementary medical device manufacturers. The facility handles formulation, filling, packaging, and quality control for the entire commercial portfolio and therapeutic pipeline.

Engineered in India, partnered globally.
Strategic partnerships with European and Australian biotech companies provide access to proprietary biological materials, international distribution channels, and regulatory expertise. Commercial B2B relationships span India, the UK, Canada, and the Middle East.

Strategic & Technology Partners
Hemarina SA
Morlaix, France
Marine haemoglobin partnership for HEMOSOME OxyRescue Serum. Arenicola marina-derived oxygen carrier for topical wound healing and aesthetic medicine applications.
Technology partnership
Holista Colltech
ASX: HCT, Australia
Exclusive supply agreement for Nano Collagen platform. ASX-listed biotech specialising in collagen extraction and nanomaterials.
Exclusive supply agreement
Commercial & Distribution Partners
Elegantes London / Harrods UK
United Kingdom
B2B DistributionSkinSeqnc Canada
Canada
B2B DistributionDefine Bio
India
SubsidiaryNano-Malaysia
Malaysia
PartnerHuman Cell Research Bharat
India
Research and Ideation ChannelAMTZ Ecosystem Partners
Visakhapatnam, India
Manufacturing EcosystemConnect with Akrivis.
Whether you are an investor, potential partner, researcher, or member of the press, we welcome the opportunity to discuss how Akrivis platforms can create value together.

IP Portfolio
Send an Inquiry
Reach out directly to the Akrivis leadership team for investment discussions, partnership opportunities, research collaborations, or media inquiries.