BIOTECH & DEEP TECH PLATFORM COMPANY · AMTZ, VISAKHAPATNAM
Advanced Therapeutics & Medical Devices.
Five platforms spanning regenerative therapeutics, advanced cosmeceuticals, and first-in-class medical devices. From concept to cGMP manufacturing at AMTZ.
"We build platforms, not products. Each product is the expression of a platform; each platform is a body of knowledge that compounds."
Five Platforms. One Integrated Vision.
Each platform represents a distinct body of knowledge — from marine biotechnology to catechol chemistry — converging into a unified therapeutic and commercial portfolio.
TympanoSeal™ NeoTympanum
Non-Invasive Tympanic Membrane Repair
A novel medical device for non-invasive tympanic membrane repair. Designed as a fifteen-minute in-clinic procedure, offering a non-surgical alternative to conventional tympanoplasty. Chronic suppurative otitis media affects approximately 65–330 million people globally (WHO, 2004), with a disproportionate burden in South and Southeast Asia.
- First-in-class to our knowledge
- Non-invasive in-clinic procedure
- Scalable manufacturing
- Large addressable patient population
SynExo™ Therapeutics
Regenerative Therapeutics Platform
The core therapeutic platform spanning wound care, burn care, and regenerative medicine. Manufactured under cGMP at the AMTZ facility in Visakhapatnam.
- Multi-source platform architecture
- cGMP manufacturing at AMTZ
- Accelerated regulatory pathway
- Broadest platform of its kind in India
EXOGLAM
Advanced Cosmeceutical Portfolio
Therapeutic and cosmetic formulations for aesthetic medicine, wound repair, and adjacent regenerative indications. European market access strategy supports international expansion.
- Dual-track: cosmeceutical + therapeutic
- European market access established
- Licensed to international partners
- Expanding global distribution
HEMOSOME OxyRescue Serum
In Partnership with Hemarina SA, France
Commercialisation of oxygenation technology developed by Hemarina SA of France. Topical applications in wound healing and aesthetic medicine.
- Exclusive partnership with Hemarina SA, France
- Unique biological mechanism
- Wound healing + aesthetic medicine
- International technology partnership
ClearSight Nano · Nano Collagen · VOC · MPTR
Nanomaterials, Diagnostics & Emerging Technologies
A portfolio of emerging platforms spanning nanomaterials, collagen technologies, non-invasive diagnostics, and next-generation therapeutic delivery systems.
- Exclusive supply agreement with Holista Colltech (ASX: HCT)
- Non-invasive diagnostic platform
- Next-generation delivery systems
- Optical nanomaterials
Development Pipeline
From validation-stage medical devices to commercial cosmeceuticals — a diversified portfolio across multiple therapeutic areas.
Therapeutic Pipeline


Commercial Portfolio
EXOGLAM Serums
Aesthetic Medicine
Approved
Nano Collagen Range
Dermal Care
Holista Colltech Partnership
HEMOSOME OxyRescue
Topical Oxygenation
Hemarina SA Partnership
SynExo™ Hair Growth
Trichology
Approved
SynExo™ Anti-Ageing
Cosmeceutical
Approved
Explore Our Technology
Interactive demonstrations of our platform capabilities and yield estimations.
Absorption Comparison
Standard Collagen vs Nano Collagen
Exclusive supply: Holista Colltech Ltd (ASX: HCT) — exclusive supply partnership. Proprietary nano-scale collagen achieving 84x greater absorption than standard collagen.
Yield Estimator
SynExo™ Production Calculator
Giga-scale production: Illustrative estimates based on Akrivis manufacturing facility at AMTZ, Visakhapatnam. Actual yields vary based on production parameters. Contact us for validated production data.
The Team
A multidisciplinary team spanning biotechnology, regulatory affairs, manufacturing, and commercial strategy.

Dr. Sarat Babu Janipalli
Founder & CEO
Two decades of experience in surgery with interests in nanomaterials and regenerative medicine. Leads platform architecture, IP strategy, and regulatory navigation across the company's therapeutic and device portfolio.

Dr. Vamshi Krishna Irlapati
Co-Founder & Chief Scientific Officer
Leads pre-clinical research, assay development, and scientific validation across all platforms. Translates research into reproducible manufacturing protocols and regulatory-grade documentation.

Mr. Ravi Eswarapu
Co-Founder & Chief Business Officer
Drives commercial strategy, international partnerships, and investor relations. Architect of the B2B distribution network spanning India, Europe, and the Middle East.

CA (Dr.) Mahaveer Chaplot
Founder — Private Equity & Strategic Finance
Prominent Chartered Accountant, educator, and community leader with over two decades of expertise in private equity, banking, and agripreneurship. Director of Agribid, an AI-driven platform empowering farmers. General Secretary of the Certified Accountants Division, International Council of Jurists. Currently serving as Sachiv (Secretary) for JITO Apex (2024–26).

Mr. Sampath Kumar Janipalli
Founder — International Operations
Manages international operations, entity coordination, and cross-border supply chain logistics. Anchors European distribution and partnership infrastructure.

Mr. Abhijit Samant
Director — Regulatory Affairs & Quality Systems
Oversees CDSCO submissions, ISO compliance, and quality management systems across the AMTZ manufacturing facility. Navigates accelerated regulatory pathways for therapeutic approvals.

Mr. Kuldeep Singh
Director — Manufacturing & Supply Chain
Leads manufacturing operations at the AMTZ facility, supply chain optimisation, and production scaling. Maintains cGMP compliance and quality standards across the commercial portfolio.
Certifications & Standards
Regulatory compliance and quality management systems underpinning every product in the portfolio.
CDSCO
Central Drugs Standard Control Organisation
Manufacturing licence for cosmeceutical and therapeutic formulations. Full commercial portfolio approved.
ISO 22716:2007
Cosmetics GMP
Good Manufacturing Practice for cosmetic products. Covers the AMTZ facility production lines for the entire cosmeceutical portfolio.
ISO 9001:2015
Quality Management Systems
Organisation-wide quality management system certification covering design, development, manufacturing, and distribution of cosmeceutical and therapeutic products.
US FDA
United States Food & Drug Administration
Facility registration and product listing for export-grade cosmeceutical formulations. Enables direct export through registered distribution channels.
BIS MHD-20
Bureau of Indian Standards — Medical & Healthcare Devices
Institutional membership on the Bureau of Indian Standards MHD-20 committee — the Indian equivalent of a CEN/CENELEC or ISO technical committee for medical devices. Akrivis participates in drafting national standards for medical and healthcare devices, analogous to how European companies contribute to EN ISO 13485 or MDR harmonised standards.
TÜV Rheinland
Independent Elemental Validation
Independent third-party validation of platform materials. Confirms elemental composition and purity across the commercial portfolio.
Manufacturing Anchor: AMTZ, Visakhapatnam
The Andhra Pradesh MedTech Zone (AMTZ) is Asia's first integrated medical device manufacturing ecosystem. Akrivis operates its cGMP facility within AMTZ, providing access to shared infrastructure, regulatory support, and a cluster of complementary medical device manufacturers.

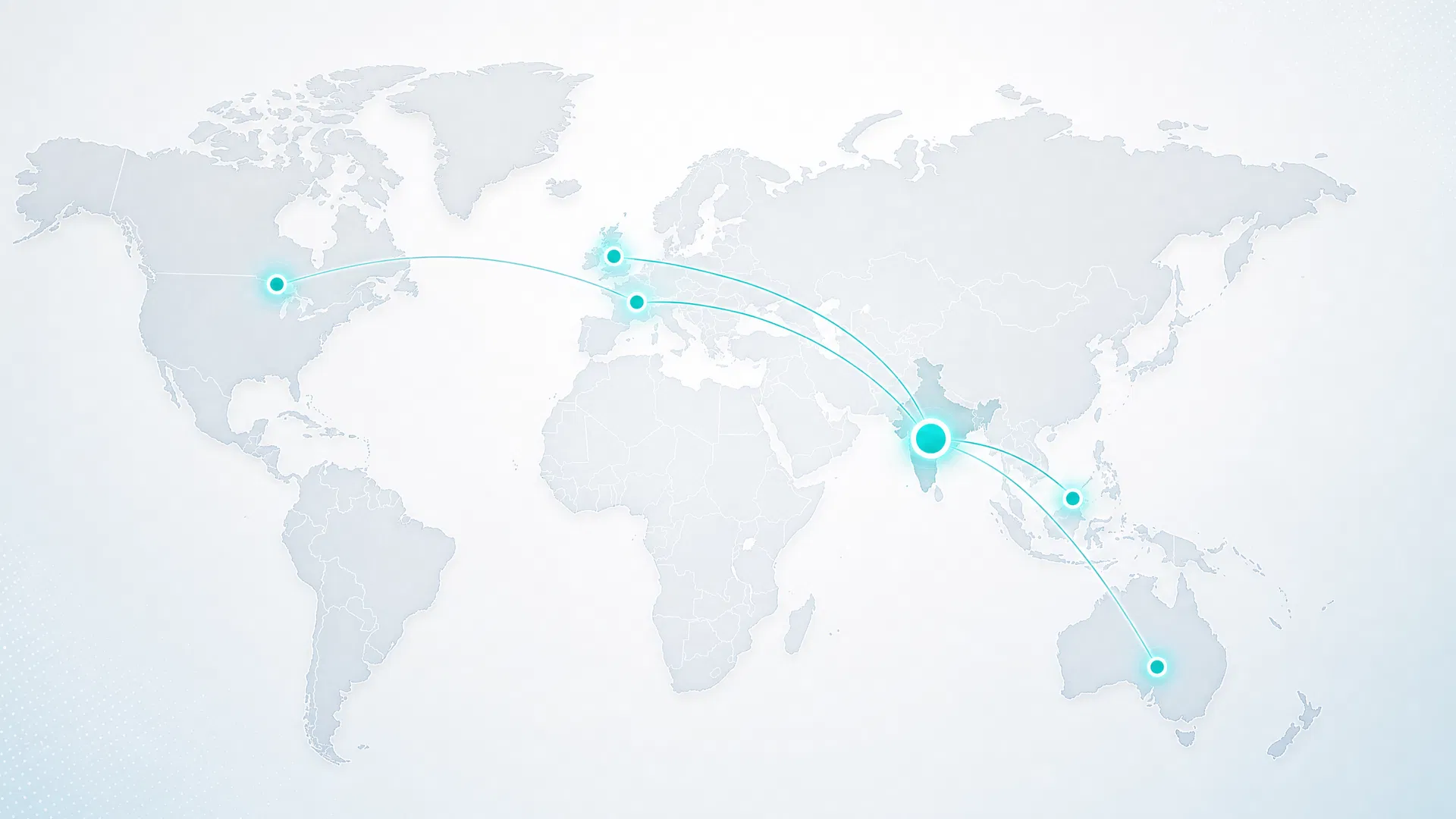
Strategic Partnerships
A curated network of technology providers, distribution partners, and institutional supporters enabling global reach from our AMTZ manufacturing base.

Technology Partners
Hemarina SA
Morlaix, France
French marine biotechnology company specialising in oxygen-carrying molecules derived from Arenicola marina. Hemarina's M101 molecule forms the basis of the HEMOSOME OxyRescue platform — a topical oxygenation technology with applications in wound healing and aesthetic medicine.
Exclusive India commercialisation rights
Holista Colltech Ltd
Kuala Lumpur, Malaysia (ASX: HCT)
ASX-listed biotechnology company specialising in nano-scale collagen extraction from ovine sources. Holista's proprietary process achieves 84× greater dermal absorption than standard collagen — forming the basis of Akrivis's Nano Collagen platform.
Exclusive supply agreement
Distribution Partners
Elegantes London / Harrods UK
United Kingdom
Premium distribution channel for the EXOGLAM cosmeceutical range through Harrods and affiliated luxury retail networks in the United Kingdom.
Distribution PartnerSkinSeqnc
Canada
Canadian distribution partner for the SynExo™ and EXOGLAM cosmeceutical portfolios. Covers the North American premium skincare market.
Distribution PartnerNano-Malaysia
Malaysia
Strategic partner for nano-collagen and cosmeceutical distribution across the Southeast Asian market.
PartnerDefine Bio
India
Subsidiary focused on domestic Indian market distribution and brand development for the cosmeceutical portfolio.
SubsidiaryIncubation & Acceleration Partners
AMTZ — Andhra Pradesh MedTech Zone
Manufacturing HubAsia's first integrated medical device manufacturing ecosystem. Akrivis operates its cGMP facility within AMTZ, providing access to shared infrastructure, regulatory support, and a cluster of complementary medical device manufacturers.
Nadupuru, Anakapalli District, Visakhapatnam, Andhra Pradesh 531019
RTIH — Ratan Tata Innovation Hub
Corporate OfficeInnovation hub providing incubation support, mentorship, and access to the broader Tata ecosystem for deep-tech startups.
IIT Hyderabad Campus, Kandi, Sangareddy, Telangana 502285
Investment Opportunity
Deep-tech biotech platform with diversified revenue streams, IP moat, and scalable manufacturing.

IP Portfolio
Send an Inquiry
Reach out directly to the Akrivis leadership team for investment discussions, partnership opportunities, research collaborations, or media inquiries.